IVF Protocol for Low Ovarian Reserve: Steps, Medications & Tips
When fertility testing shows a low number of eggs remaining, IVF can still be an option but the strategy needs to be modified. “Low ovarian reserve” means the ovaries are likely to produce fewer eggs with stimulation than expected for age. It can occur at any age and is more common as we get older.
The goal of a good protocol isn’t to “force” the ovaries to make lots of eggs, but to help them yield the best-possible few, then protect every step that follows (fertilization, embryo development, transfer timing).
In this guide, we’ll translate the evidence behind different stimulation approaches, add-ons, and planning choices. We’ll also clarify how low ovarian reserve differs from primary ovarian insufficiency (POI), what success realistically looks like, and how to think about costs in India.
Difference Between “Low Ovarian Reserve” vs “Poor Responder”
The Bologna criteria (2011) created a common language for “poor ovarian response,” typically requiring at least two of: advanced maternal age (≥40) or risk, previous poor response, and abnormal ovarian reserve tests (low AMH/AFC).
The newer POSEIDON framework classifies “low-prognosis” patients by age, ovarian reserve markers, and observed response to stimulation, to guide individualized planning. These are used for counseling and study design.
AMH and AFC help anticipate response (how many follicles might grow). AMH <˜1 ng/mL often signals reduced response; AMH <0.5 ng/mL is frequently associated with ≤3 follicles, while typical values fall with age (about ˜2 at 30, ˜1.5 at 35, ˜1 at 40). AFC and AMH are the most informative routine predictors for response to stimulation. These tests do not guarantee or rule out pregnancy on their own.
Low Reserve vs POI
Low reserve means fewer recruitable follicles but periods may be normal. Primary ovarian insufficiency (POI) involves loss of ovarian function before 40, often with irregular or absent periods. Some people with POI still intermittently ovulate and can conceive, but management and expectations differ.
Signs & Symptoms of Low Ovarian Reserve (LOR)
Most people with LOR don’t feel “sick.” The clues are subtle and often turn up during fertility work-ups:
- Trouble conceiving for 6–12 months despite regular cycles
- Shorter menstrual cycles (for example, 24–26 days instead of ˜28) and/or lighter flow
- Lab/scan findings like low AMH, high day-2/3 FSH or estradiol, and a low antral follicle count (AFC) on ultrasound
Causes & Risk Factors
Age is the biggest driver, but LOR can occur earlier for other reasons:
- Ovarian reserve naturally declines with time
- Genetic factors/family history. Some families experience earlier menopause; fragile-X premutation is one known association
- Endometriosis or prior ovarian surgery as cyst removal can reduce follicle pool
- Chemo/radiation or pelvic infection can cause direct ovarian damage
- Lifestyle/environment factors like smoking; possible environmental toxins
- Autoimmune/idiopathic as sometimes no clear cause is found
How LOR Is Diagnosed (and Distinguished from “Poor Response”)
Clinics combine clinical context with tests:
- AMH: typically low (<˜1.0 ng/mL on many assays; exact thresholds are assay-specific)
- Day-2/3 FSH and estradiol: FSH may be elevated; estradiol can “mask” FSH
- AFC on transvaginal ultrasound: fewer small follicles (often <5–7, clinic-dependent)
Optional tests: prior cycle response, clomiphene challenge in select cases
Treatment Procedure: Step-by-Step Pathway for IVF in Low Ovarian Reserve
IVF for LOR focuses on recruiting the best possible techniques with the least suppression, retrieving whatever eggs the ovary can offer, and maximizing embryo potential over one or more cycles.
1) Pre-cycle preparation & counseling
Review age, prior responses, AMH/AFC, and co-morbidities; set realistic goals (often cumulative live birth over multiple cycles).
Discuss adjuvants: CoQ10 and DHEA are commonly considered; evidence is mixed to moderate and varies by age. Use only under specialist guidance.
2) Choose a stimulation strategy that avoids over-suppression
Common, evidence-based options your clinician may consider:
Higher gonadotropin DosesBeyond a point, simply increasing the dose often yields diminishing returns. Many clinics escalate to 300–450 IU/day FSH after a weak response. However, reviews suggest that very high doses rarely transform outcomes and can increase cost and burden. Individualization is better than a one-size “maximum dose for all.”
rFSH alone vs adding LH activity (rLH or low-dose hCG)Adding LH activity may benefit selected poor responders, but evidence is mixed. Several large retrospective studies and reviews indicate that recombinant LH (rLH) supplementation can improve cumulative outcomes in moderate–severe poor responders.
Low-dose hCG during stimulation (as a surrogate for LH activity) has shown improved follicle maturity and embryo quality in some trials and may be cost-effective, but results vary and need careful candidate selection.
Antagonist vs Agonist ProtocolsModern GnRH antagonist cycles are popular for poor responders because they’re shorter, require fewer injections, and avoid deep ovarian suppression. Antagonists reduce the risk of premature LH surge while allowing flexibility to add LH or letrozole, and to pivot to freeze-all if needed. Agonist “flare” and “stop” regimens remain options for selected cases but show inconsistent advantages. Choice is usually based on your prior cycle data and clinic experience.
Mild Stimulation vs Conventional “High-Dose” StimulationMild stimulation (≤150 IU/day with or without oral agents) can offer similar cumulative outcomes in low-prognosis patients at lower cost and burden; live-birth rates per cycle remain modest either way.
Randomized and cohort studies suggest comparable cumulative live birth between mild and conventional dosing in low-prognosis groups, acknowledging that conventional dosing often yields more oocytes. Many guidelines encourage considering mild protocols in poor responders, especially after a weak high-dose cycle.
Natural-Cycle IVF (No Stimulation) Reasonable in select scenarios, but typically limited in poor responders because one egg means one roll of the dice. A recent review concludes that natural cycles have limited potential in poor responders, though individual clinics may use modified natural strategies to avoid over-medication or when stimulation repeatedly fails.
Key technical point: The objective is not to “force” a high egg number with very high FSH (which often adds cost with little gain in LOR), but to optimize recruitment and oocyte competence. (Multiple studies found limited benefit to simply escalating gonadotropin dose.)
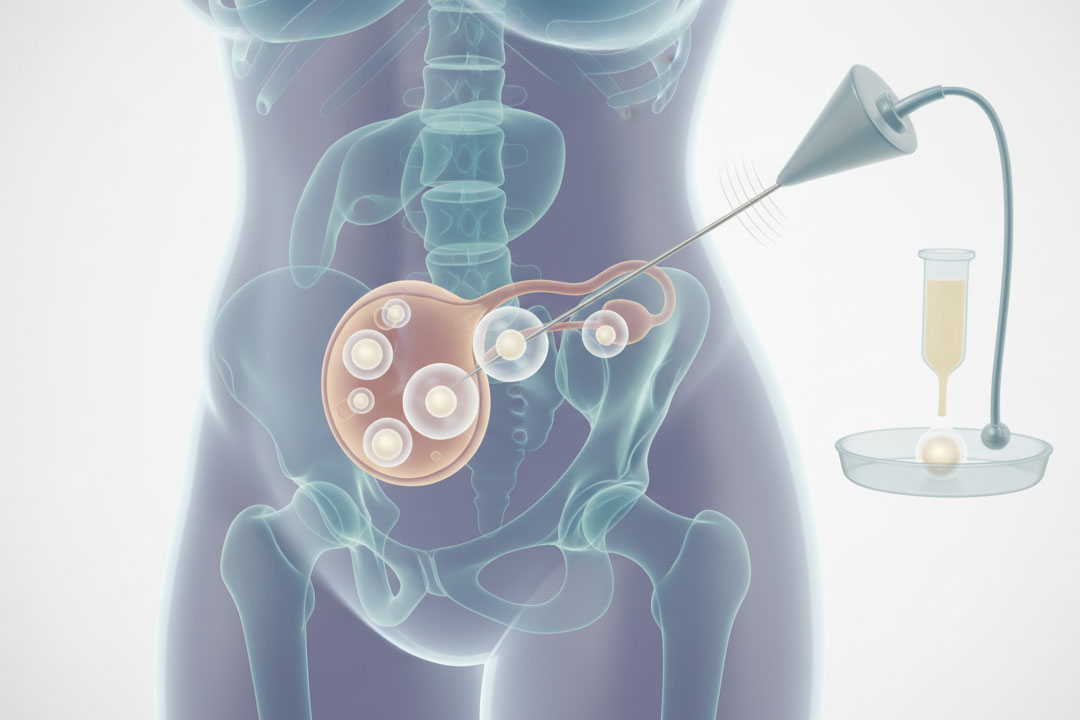
3) Monitoring, trigger, and retrieval
Close ultrasound/estradiol monitoring to avoid premature LH surge and time the trigger precisely.
Trigger with hCG or a dual trigger (GnRH-a + low-dose hCG) depending on protocol and OHSS risk (usually low in LOR). Egg retrieval ˜34–36 hours after trigger.
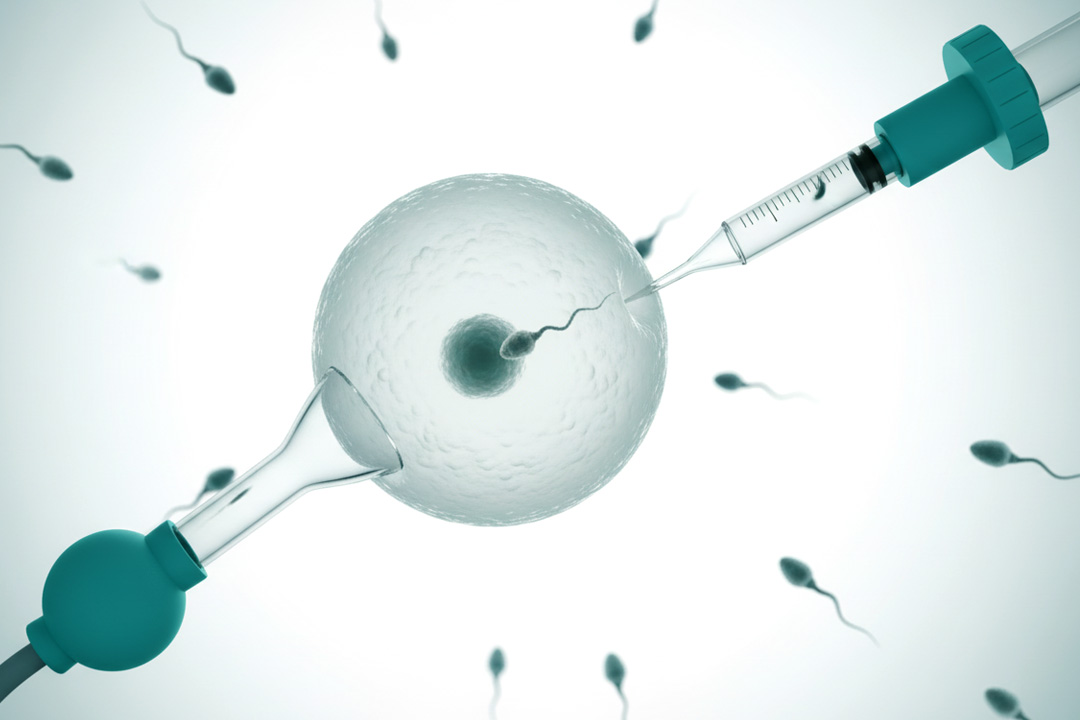
4) Fertilization & Lab Strategy
ICSI is often used in LOR to minimize fertilization failure (especially with few oocytes). Consider embryo culture to blastocyst when feasible; however, when oocyte numbers are very low, some clinics individualize day-3 transfer decisions.
5) Transfer strategy, “freeze-all,” and embryo pooling
Historically, freeze-all showed advantages in some populations (lower OHSS; sometimes higher ongoing pregnancy). Newer evidence suggests that fresh transfer may outperform freeze-all specifically in low-prognosis women, so this choice should be individualized based on endometrium, hormone levels, and embryo number/quality.
Embryo pooling (accumulation) across two or more retrievals can raise the chance of having at least one high-quality embryo to transfer, which matters when each retrieval yields few oocytes.
6) Luteal support and beyond
Progesterone support is standard after retrieval/transfer.
If cycle outcomes are sub-optimal, debrief using the POSEIDON framework to adjust dose, add LH activity, consider adjuvants, or plan pooling. BioMed Central
7) Alternatives if response remains very low
Donor oocytes or embryo donation (highest success rates) or “Ovarian rejuvenation” with platelet-rich plasma (PRP)
What it is: Intra-ovarian injection of autologous PRP before IVF. Some studies report improved AFC or oocyte numbers, while others show no significant benefit on euploidy or pregnancy. Current systematic reviews call for better trials; if offered, it should be framed as investigational with uncertain benefit.
Beyond Stimulation: Lab and Transfer Strategy Matter
Freeze-all allows better endometrial preparation and PGT-A (if chosen), and is frequently used in low-reserve cases to maximize each embryo’s chance. Poor responders often create few embryos; banking them across cycles (“embryo pooling”) can allow the best embryo to be transferred later and, if desired, enable PGT-A to avoid aneuploid transfers.
Costs in India
Costs vary by city and clinic, but typical ranges (procedure with most meds) are around ₹1,50,000–₹3,00,000 per cycle; medications can add substantially depending on dose and protocol. Add-ons (GH, PGT-A, PRP) increase costs and should be weighed against evidence. Always request an itemized estimate.
Frequently Asked Questions
Is IVF for me if my AMH is low at 30?
Yes, age is a strong positive factor. Even with low reserve, people ≤35 often do reasonably well when protocols are tailored and embryos are pooled if needed. AMH helps predict response, not whether you can get pregnant. Discuss a mild or moderate antagonist plan, with or without letrozole, and consider banking embryos over 1–2 cycles before transfer.
Is mild stimulation really enough?
For low-prognosis patients, mild stimulation can deliver similar cumulative outcomes to high-dose plans at lower medication burden and cost. It’s not universally best, but it’s a valid first-line or second-line option, especially if high-dose cycles are under-performed.
Should I add LH (rLH) or low-dose hCG?
Maybe. If prior cycles showed poor maturation or low estradiol despite adequate FSH, adding LH activity can help some patients. Evidence is mixed; benefits appear strongest in specific subgroups. Your earlier cycle data guide this choice.
Do supplements like DHEA, testosterone gel, or GH improve live birth?
It is unclear. DHEA and testosterone data are inconsistent; GH shows low-certainty evidence on live birth. If used, they should be time-limited, with informed consent about uncertain benefit versus added cost. CoQ10 has encouraging data on response and embryo quality with a good safety profile and is commonly tried pre-cycle.
What about PRP “ovarian rejuvenation”?
Experimental. Some studies show higher AFC or oocyte numbers; others show no improvement in euploidy or pregnancy. Consider only in research settings or after detailed counseling.
Is “natural-cycle IVF” advisable for me?
It can be considered if you strongly prefer minimal meds or have repeatedly over-suppressed, but reviews suggest limited potential in typical poor responders. A modified natural approach is sometimes used case-by-case.
Should I transfer fresh or freeze them all?
Many low-reserve patients benefit from freeze-all to optimize endometrium, time the transfer, and optionally add PGT-A. Others may transfer fresh if embryo numbers are very low and the lining looks good. Your lab’s outcomes and your priorities drive the decision.
Conclusion
Low ovarian reserve doesn’t close the door on parenthood but it changes how we plan the journey. The most effective IVF care here is individualized: gentle or antagonist-based stimulation, timely LH activity, and selective use of add-ons (such as letrozole or growth hormone) to prioritize egg and embryo quality over sheer numbers.
Lifestyle foundations and clinic-guided supplements may support the plan; donor options remain a compassionate alternative if response stays very limited. What matters most is aligning expectations with a clear, stepwise strategy aimed at cumulative live birth. With a skilled team and a tailored protocol, many patients with low reserve still achieve the outcome they’re hoping for.
About Us
AKsigen IVF is a premier center for advanced fertility treatments, with renowned fertility experts on our team. Specializing in IVF, ICSI, egg freezing, and other cutting-edge reproductive technologies, AKsigen IVF is committed to helping couples achieve their dream of parenthood. With personalized care and a patient-first approach, AKsigen IVF provides comprehensive fertility solutions under one roof.